Measuring Ozone
The PocketLab Air ozone sensor measures ozone concentration through an electrochemical reaction.
Background information about ozone
Ozone (O3) in our atmosphere is both good and bad. There is a helpful saying to remember is, “Ozone: good up high, bad nearby.” The “good” ozone is ozone high in our atmosphere that is part of the ozone layer. The ozone layer protects us from the sun’s ultraviolet rays. “Bad” ozone is ozone that occurs at ground level, where it can be inhaled. Ground level ozone is a pollutant and creates smog.
Ground level ozone is not directly emitted. It is the result of sunlight causing a chemical reaction between nitrogen oxide (NOx) and volatile organic compounds (VOC). NOx and VOCs are emitted directly from sources like chemical plants, refineries, and cars. Ground level ozone can cause a variety of health problems and can weaken plants and ecosystems.

Image from the US EPA
How does the PocketLab Air measure ozone?
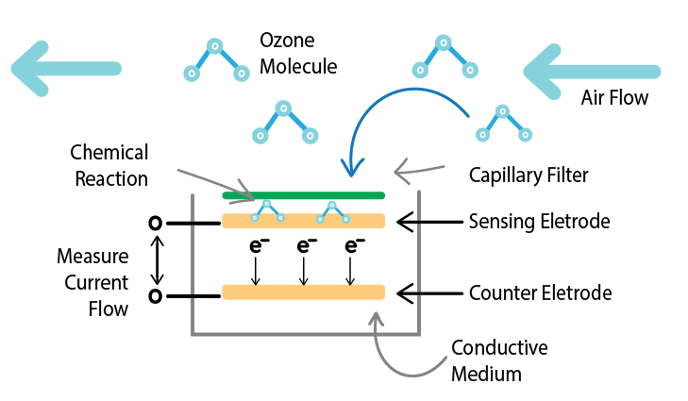
Typical electrochemical sensors are large industrial sensors that use a liquid electrolyte. The PocketLab Air uses a new type of sensor with a solid state electrolyte.
The electrochemical ozone sensor operates through the following method:
- Ozone in the air passes through a filter to screen out particles and moisture.
- The ozone molecules undergo a chemical reaction with the sensing electrode that generates free electrons.
- The electrons flow through a conductive medium to the counter electrode.
- The flow of electrons creates an electric current that is proportional to the concentration of ozone gas.
- The electrodes are connected to a circuit to measure and amplify the current signal.